 In the ring structures of sucrose and maltose, you have an anomeric carbon. The reducing ability of disaccharides is defined by the presence of a potential aldehyde or ketone group. Maltose is made up of two glucose units while sucrose is made up of glucose and fructose. Why sucrose is a non-reducing sugar but maltose is not? Note: Monosaccharides are considered as reducing sugars. Lactose undergoes mutarotation and it is hence a reducing sugar. 
Lactose is composed by the combination of a glucose and a galactose molecule. Maltose is a reducing sugar, whereas sucrose is a non-reducing sugar. The end of the molecule containing the free anomeric carbon is called the reducing end, and the other end is called the nonreducing end. What is the difference between a reducing and a non reducing end of a chain? In basic aqueous media, nonreducing sugars do not generate any compounds containing an aldehyde group. Some disaccharides and all polysaccharides are reducing sugars. Nonreducing sugars are carbohydrates that cannot act as reducing agents due to the absence of free aldehyde groups or free ketone groups. What is the difference between reducing sugars non reducing sugars and polysaccharides? Hence starch is considered to be a non-reducing sugar. Starch does not feature a free aldehyde or keto group. What carbohydrates are non-reducing?Ī non-reducing sugar is a carbohydrate that is not oxidized by a weak oxidizing agent (an oxidizing agent that oxidizes aldehydes but not alcohols, such as the Tollen’s reagent) in basic aqueous solution. Both monosaccharide anomeric carbons are implicated in glycoside or acetal production. How do you tell if a sugar is reducing or nonreducing?Īnswer: Sucrose is a non-reducing sugar since it does not dissolve in water. Disaccharides are formed from two monosaccharides and can be classified as either reducing or nonreducing. The common dietary monosaccharides galactose, glucose and fructose are all reducing sugars. 
Sucrose is a non-reducing sugar because the monosaccharides’ anomeric carbon is involved in glycoside or acetal formation. Why fructose is non-reducing sugar?Ī) Lactose b) Maltose c) Sucrose d) Fructose. Significant levels of trehalose in plants act as protectants against various abiotic stresses, including heat, drought, high salinity, and UV rays. Trehalose is a nonreducing sugar commonly found in bacteria, fungi, yeast, insects, and plants. Since the reducing groups of glucose and fructose are involved in glycosidic bond formation, sucrose is a non-reducing sugar. Sucrose is a non-reducing sugar because The two monosaccharide units are held together by a glycosidic linkage between C1 of α-glucose and C2 of β-fructose. Why sucrose is called non-reducing sugar? The reducing sugar can reduce the capric ions of the Fehling or the Benedict solution into the cuprous ions whereas, the reduction of cupric ions into the cuprous ions is not achieved in the non-reducing sugars. The reducing sugars are mainly monosaccharides where all polysaccharides are non-reducing sugars. What is the difference between reducing and non-reducing sugar a level biology? 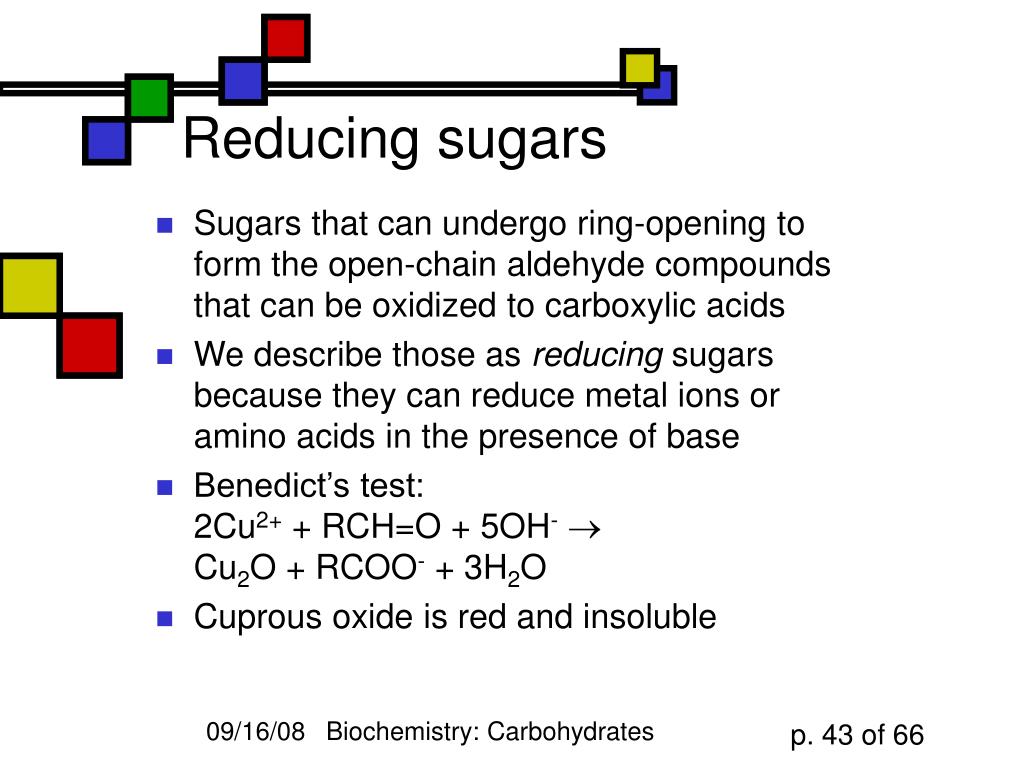
All monosaccharides such as glucose are reducing sugars. Non-reducing sugars do not have an OH group attached to the anomeric carbon so they cannot reduce other compounds. 
Reducing sugars are sugars where the anomeric carbon has an OH group attached that can reduce other compounds. What is the difference between reducing and non-reducing sugars? What are reducing sugars in biology?Ī reducing sugar is a carbohydrate that is oxidized by a weak oxidizing agent (an oxidizing agent capable of oxidizing aldehydes but not alcohols, such as the Tollen’s reagent) in basic aqueous solution. To identify a non-reducing sugar in a sample, it must be hydrolyzed first to break disaccharides into two monosaccharides before carrying out Benedict’s test. Since non-reducing sugars do not possess the ability to donate electrons, hence they cannot be oxidized. What is a non reducing sugar a level biology? Carbohydrate: A carbohydrate can be defined as a biomolecule that owns the atoms of hydrogen, oxygen and carbon. Stachyose, Sucrose, Verbascose, Trehalose and Raffinose are the five examples of non reducing sugars. 24 Is glycogen reducing or non reducing sugar? What is non reducing sugar and example? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
